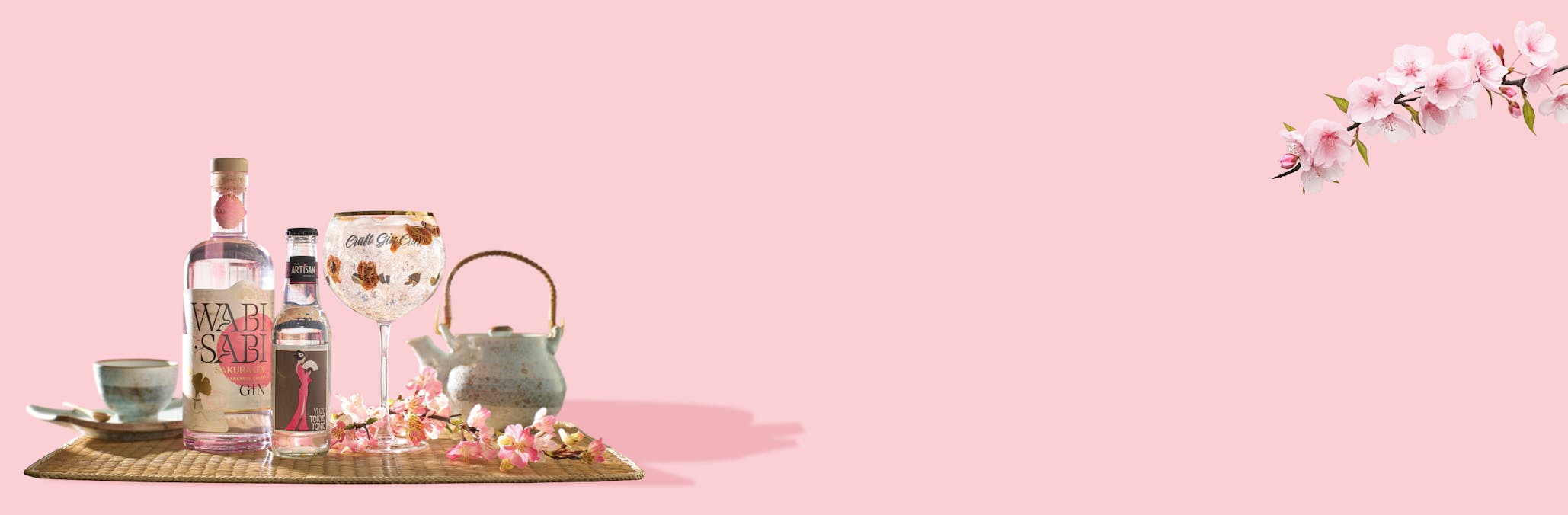Total flexibility, no commitment
A world of unique, crafted gins
Easy, free and reliable delivery

Why Gin and Tonics taste amazing - according to science!
We've always known that a G&T is greater than the sum of its parts, but now science has explained why...
According to Matthew Hartings, an assistant professor of chemistry at American University and co-author of the ScienceGeist blog, we have molecular chemistry to thank for the greatness of the G&T.
It all comes down to the shape of the molecules that make up your tipple. As it happens, the essential oils that flavour gin – primarily juniper – are very similar in shape on a molecular level to quinine, the primary flavour in tonic water.
Molecules are attracted to other molecules that look like them – so, when you mix gin and tonic, molecules with similar shapes will make a beeline for each other. This is illustrated in the figure below; the blue hexagonal molecules in the gin will be attracted to their counterparts in the tonic, as will the molecules in red.
Hartings explains: “The parts of these molecules that look alike are attracted to one another. When they are mixed together in a gin and tonic, the molecules come together to create an aggregate.”
As quinine and juniper molecules nestle together, the flavour they produce changes.
Hartings says: “This is why a gin and tonic doesn't taste exactly like gin plus tonic.”
This principle of molecular chemistry is behind all of our favourite food pairings – and some not-so-common ones, like oyster and kiwi or pork liver and jasmine. Both are probably amazing, but we might stick with the G&Ts for now.